| A new hope against pathogenic bacteria |
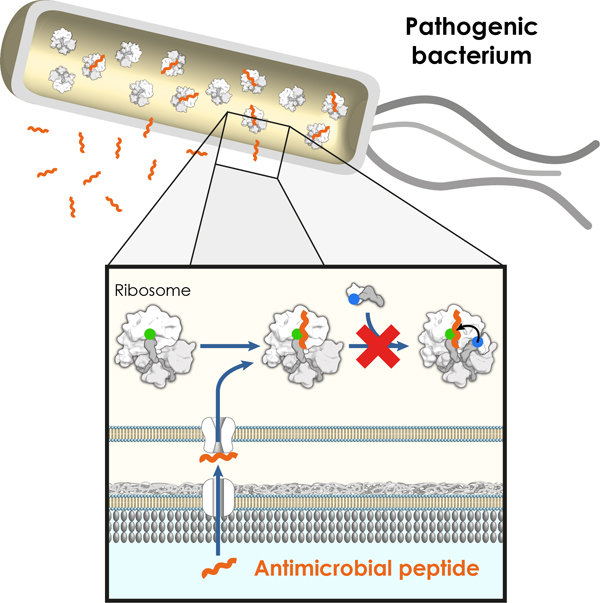
A new hope against pathogenic bacteria Two teams of the IECB collaborating with a research team from the University of Munich have revealed how insect-derived antimicrobial peptides target the protein synthesizing machinery in bacteria. These results may in turn lay the foundations for the development of novel or improved antibiotics and have just been released in the journal Nature Structural and Molecular Biology. The team of Dr. Axel Innis, in collaboration with the teams of Dr. Gilles Guichard and Dr. Daniel Wilson, has revealed how certain peptides produced by insects kill pathogenic bacteria by targeting their ribosomes, the molecular factories that produce all of the proteins needed for their survival. Though they had been poorly characterized until recently, such antimicrobial peptides could now pave the way for effective new treatments against bacteria that are resistant to current therapies.
These studies showed that oncocin interacts with a long ribosomal tunnel through which newly synthesized proteins travel to reach the intracellular milieu. Once anchored in the tunnel, the peptide masks the active site of the ribosome, thereby preventing the bacteria from synthesizing new proteins. The site of interaction between the tunnel and oncocin overlaps with regions of the ribosome that are targeted by many antibiotics, emphasizing the importance of these findings for the development of future antimicrobial compounds.
*: Thermus thermophilus is a Gram-negative bacterium. Pathogenic Gram-negative bacteria (e.g. Klebsiella, Pseudomonas or Acinetobacter) are a major problem for hospitals, nursing homes and other healthcare institutions, where they cause often difficult to treat infections in the elderly or the seriously ill. |
2, Rue Robert Escarpit - 33607 PESSAC - France
Tel. : +33 (5) 40 00 30 38 - Fax. : +33 (5) 40 00 30 68