
Dr. Axel Innis
Inserm (CR1)
This e-mail address is being protected from spambots. You need JavaScript enabled to view it
Tel : +33 (0)5 4000 6149
http://innislab.org/
Bio
Axel Innis did his PhD in structural biology at the University of Cambridge, under the supervision of Prof. Sir Tom Blundell (1998-2002). He then joined the group of Dr. R. Sowdhamini at the National Centre for Biological Sciences in Bangalore as a visiting fellow (2002-2004), where he developed a computational method for identifying functionally important sites in proteins. Following his time in India, Dr. Innis joined the laboratory of Prof. Thomas A. Steitz at Yale University (2004-2012). There, he chose to tackle what was, at the time, a little-known form of translational control: the regulation of ribosomal protein synthesis by the nascent polypeptide. He joined IECB as a group leader in January 2013 and was recruited as an Inserm research officer (CR-1) the same year.
Summary
Ribosomes are the large macromolecular complexes responsible for translating genetic information contained within a messenger RNA (mRNA) into protein in all living organisms. As part of the process of translation, nascent polypeptides transit through a long molecular cavity spanning the large subunit of the ribosome – known as the exit tunnel – before they are released into the cytoplasm or delivered to the protein translocation machinery.
Our lab uses combined biochemical, structural and computational approaches to study some of the key events that take place within the functional nano-environment of the exit tunnel, including:
- Peptide bond formation, which is catalyzed by the peptidyl transferase center located at the tunnel entrance.
- Nascent chain-mediated translational arrest, a process whereby signals encoded in certain nascent polypeptides termed arrest peptides bring protein synthesis to a halt.
- Translation inhibition by antimicrobial peptides or antibiotics that target the exit tunnel and the peptidyl transferase center of the ribosome.
Activity report
Peptide Bond formation
In order to understand how peptides or antibiotics inhibit peptide bond formation, we must first have a clear picture of the mechanism by which ribosomes catalyze peptidyl transfer. Peptide bond formation takes place within an active site that is composed primarily of RNA. Our high-resolution structures of the bacterial ribosome in complex with full-length tRNA substrates reveal a network of hydrogen bonds (or “proton wire”) along which proton transfer could take place to assist catalysis (Polikanov et al., 2014). This has led us to propose a mechanism for peptide bond formation in which the ribosome together with the A- and P-tRNAs trigger the reaction by activating a water molecule (Fig. 1). As this proposed catalytic water is cut off from the bulk solvent by the N-terminus of ribosomal protein L27 in bacteria, we are currently investigating a possible regulatory role for this protein during translation.
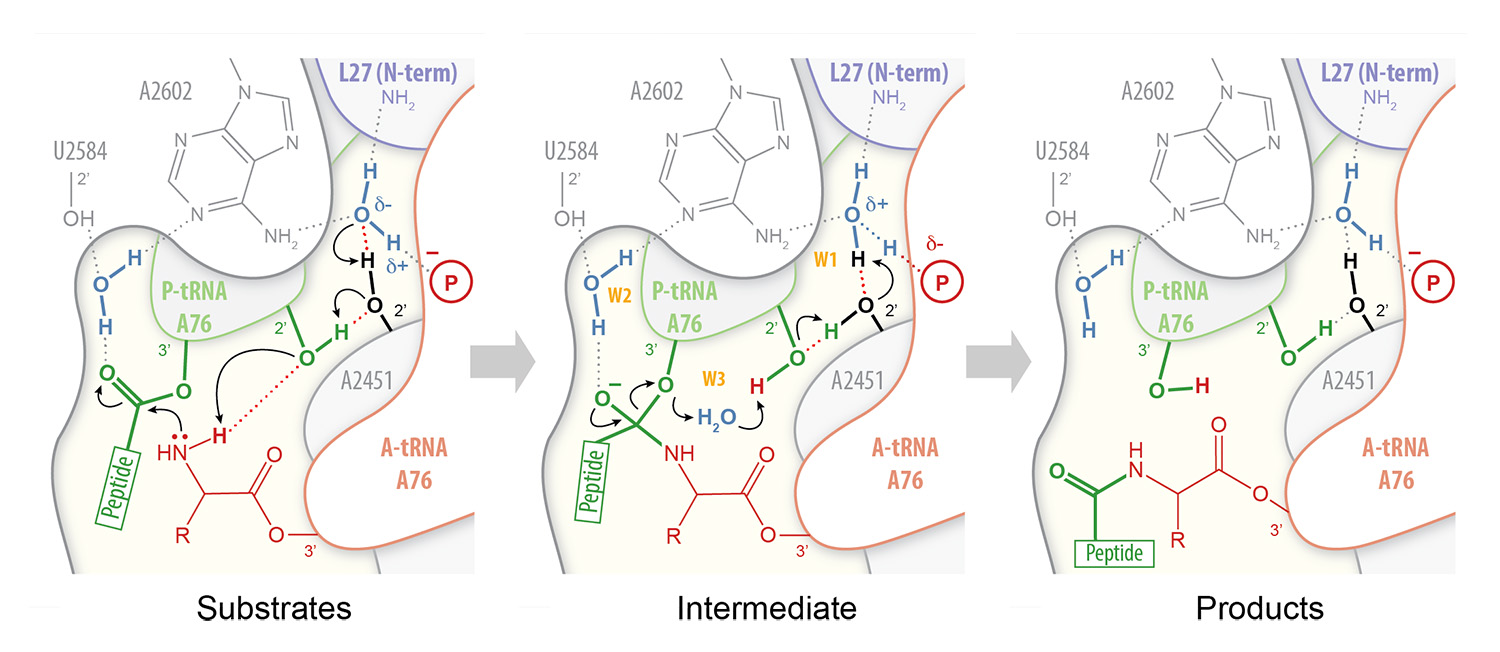
Figure 1 – Proton wire for peptide bond formation. In our proposed mechanism for peptide bond formation, nucleophilic attack is facilitated through the deprotonation of the a-amine of the incoming amino acid by a catalytic water molecule (W1) positioned at the extremity of a “proton wire” (Polikanov et al. 2014).
Nascent chain-mediated translational arrest
Translation inhibition by arrest peptides is critically dependent on their amino acid sequence, but often requires an additional low molecular weight ligand, such as a drug or a metabolite, to be sensed by the ribosome nascent chain complex. Thus, arrest peptides are used for metabolite-dependent gene regulation in both prokaryotes and eukaryotes. Biological processes that are regulated by arrest peptides in bacteria include the induction of erm resistance genes by macrolide antibiotics (e.g. erythromycin), the sensing of soluble tryptophan by a ribosome-associated TnaC peptide, targeting of the expression of the SecA pre-protein translocase to the cell membrane by the nascent SecM polypeptide, the expression of the YidC2 membrane insertase by the MifM peptide (Sohmen et al. 2015) and the regulation of SecDF2 in low-salinity environments by the arrest peptide VemP.
Biochemical and structural studies have shown that interactions between nascent peptides and the ribosome that induce translational arrest do so by impairing tRNA accommodation, peptide bond formation or peptide release. However, the arrest code dictating whether a given nascent peptide is prone to inhibiting its own synthesis is yet to be elucidated, the range of metabolites that can be sensed by the nascent peptide is unknown and the molecular bases of the arrest mechanism itself are only partially understood. As a result, we are developing new high-throughput tools to systematically address these issues on an unprecedented scale.
Antimicrobial peptides
The threat posed by multidrug-resistant bacteria presents a major public health challenge that requires immediate and coordinated action on a global scale. The bacterial ribosome is a major target for antibiotics, many of which bind to the exit tunnel. This includes drugs that inhibit peptide bond formation (e.g. chloramphenicol), as well as compounds that selectively interfere with the movement of the nascent peptide down the exit tunnel (e.g. erythromycin).
In addition, we have recently shown that proline-rich antimicrobial peptides (PrAMPs) produced by the host immune response of insects and mammals inhibit translation by blocking the exit tunnel and peptidyl transferase center of the ribosome (Seefeldt et al. 2015; Seefeldt et al. 2016) (Fig. 2). These natural compounds share structural similarities with arrest peptides, indicating that the latter could help steer the search for new peptide-based antimicrobials that are effective against antibiotic-resistant pathogens.
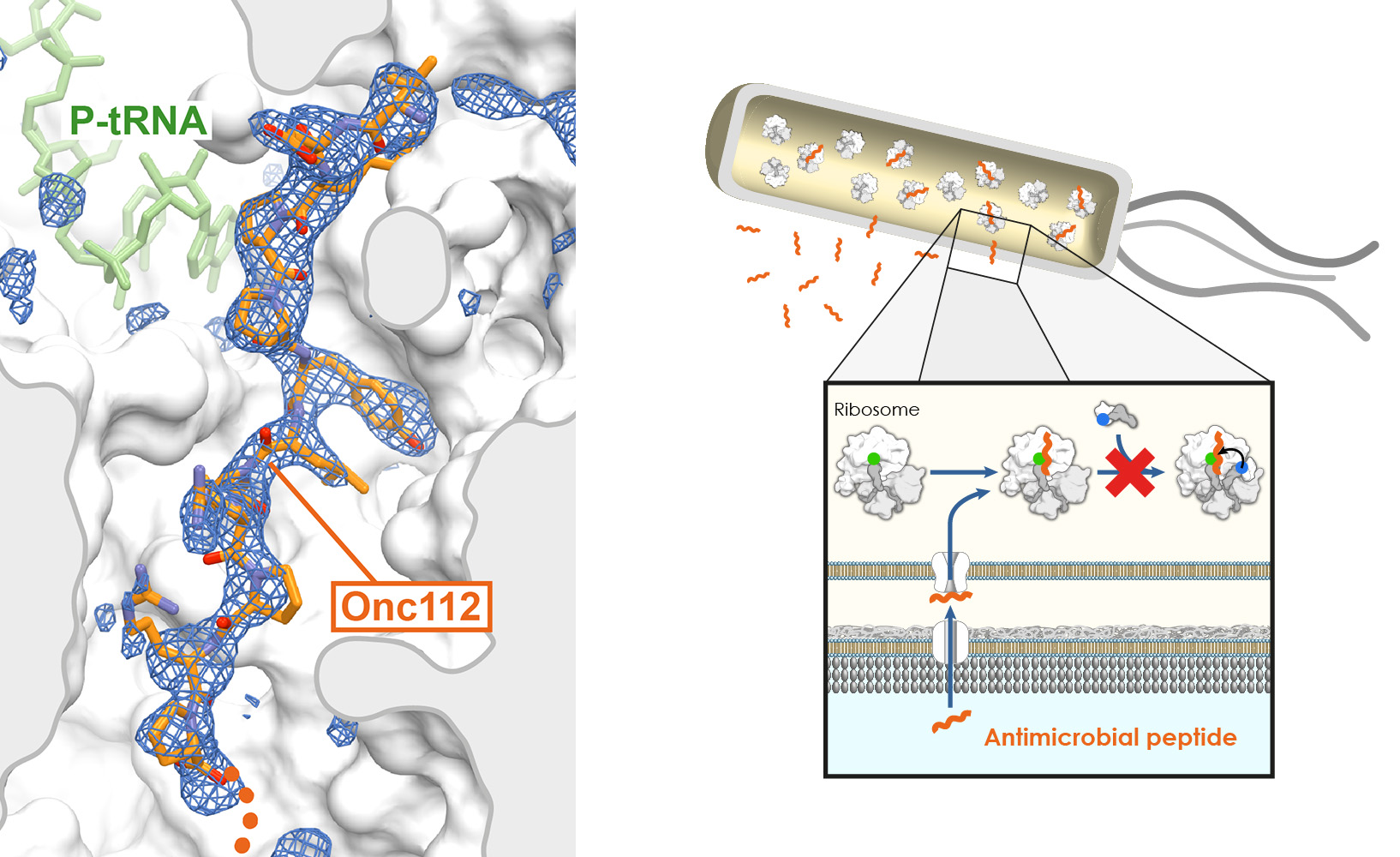
Figure 2 – Ribosome inhibition by antimicrobial peptides. The insect-derived proline-rich antimicrobial peptide Onc112 inhibits bacterial protein synthesis by blocking and destabilizing the translation initiation complex (Seefeldt et al. 2015). Other PrAMPs like Bac7, Metalnikowin or Pyrrhocoricin operate through a similar mechanism (Seefeldt et al. 2016).
Selected publications
- Seefeldt, A.C., Graf, M., Nguyen, F., Pérébaskine, N., Arenz, S., Mardirossian, M., Scocchi, M., Wilson, D.N., Innis, C.A.†(2016). Structure of the mammalian antimicrobial peptide Bac7(1-16) bound within the exit tunnel of a bacterial ribosome. Nucleic Acids Res. 44, 2429-2438.
- Seefeldt, A.C., Nguyen, F., Antunes, S., Pérébaskine, N., Graf, M., Arenz, S., Inampudi, K.K., Douat, C., Guichard, G., Wilson, D.N., Innis, C.A.†(2015). The proline-rich antimicrobial peptide Onc112 inhibits translation by blocking and destabilizing the initiation complex. Nat. Struct. Mol. Biol. 22, 470-475.
- Sohmen D, Chiba S, Shimokawa-Chiba N, Innis CA, Berninghausen O, Beckmann R, Ito K, Wilson DN. (2015). Structure of the Bacillus subtilis 70S ribosome reveals the basis for species specific stalling. Nat. Commun. 6, 6941.
- Polikanov, Y.S., Steitz, T.A., Innis, C.A.†(2014). A proton wire to couple aminoacyl-tRNA accommodation and peptide bond formation on the ribosome. Nat. Struct. Mol. Biol. 21, 787-793.
Research Team
- Dr. Axel INNIS Team Leader (Inserm)
- Dr. Britta SEIP, Post-doc (University of Bordeaux)
-
Dr. Justine CHARON, Post-doc (Inserm)
- Natacha PEREBASKINE, Technician (University of Bordeaux)
- Carolin SEEFELDT, PhD Student (Inserm)
- Alba HERRERO DEL VALLE, PhD Student (University of Bordeaux)
-
Guénaël SACHEAU, Master student (University of Bordeaux)
This team is part of the unit "ARN : RĂ©gulations naturelle et artificielle" (ARNA), Inserm
U1212 – CNRS UMR 5320/Université de Bordeaux
|

 Research and training
Research and training




