
Dr. Michel Laguerre
Directeur de recherche (DR2), CNRS
Cette adresse email est protĂŠgĂŠe contre les robots des spammeurs, vous devez activer Javascript pour la voir.
Website
Tel: +33(0)540002212
Â
Bio
Michel Laguerre, graduate from the Ecole Nationale SupĂŠrieure de Chimie of Toulouse, obtained his Engineering thesis in 1977 and his State Thesis (DSC) in Chemistry (UniversitĂŠ Bordeaux I) in 1979 under the supervision of Raymond Calas (Organosilicon chemistry). He was hired by the CNRS in 1980 and joined the Life Sciences Department of the University Bordeaux Segalen, where he worked on the synthesis and design of drugs in the central nervous system area. In 1994 he moved to Centre de Recherche Paul Pascal (CRPP) in the Chemistry Department of CNRS where he reorientated his research axis toward biomembrane models and lipidic assemblies. After being promoted Directeur de recherche, he joined the IECB in 1998.
Â
Keywords / Expertise / Techniques
molecular modeling and molecular dynamics, lipidic assemblies, biomembrane models, mono- and bi-layers, micelles, membrane proteins, GPCR, building of protein models by sequence homology, apoptotic cascade, kinases and drug-design, mesoscopic or coarse-grain dynamics.
Â
Summary
Due to the increasing power of available computers, molecular simulation is now becoming an invaluable tool for structural biology. Using an all-atom representation, molecular dynamics allows a deep insight into the behavior of biomolecules. This approach is used in the context of three axes of research : lipidic assemblies, proteins and finally membrane proteins within biomembrane models. To overcome the limitations of the all-atom approach, mesoscopic representations of lipidic assemblies or proteins are developped, so as to gain access to simulations on long time or space scales. Finally in silico drug-design techniques allow to fulfil some gaps at the medicinal chemistry interface.
Â
Activity report
All-atom molecular dynamics
Our first research axis is devoted to molecular dynamics of complex lipidic assemblies using an all-atom representation : i.e., spherical or cylindrical micelles of various surfactants, Langmuir films and various bilayers of biologically relevant lipids. This work is performed in close collaboration with several teams involved in experimental biophysics. Very recently, we succeeded in determining at the atomic level the global structure of a nano-object containing tartrates of geminis. This is the first structure at an atomic level of such a nano object (published in JACS).
Â
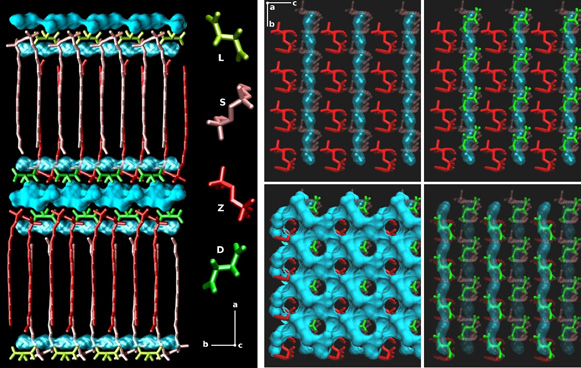
Figure 1: Final structure of the gemini tartrate film structure at an atomic level: geminis are in shade of red, tartrates in shade of green and water molecules are in blue. Tubes of aligned water molecules can be easily seen in the right panel. Between the two interdigitated layers a hexagonal lattice of water molecules insures the global cohesion of the structure.
Concerning the protein axis, we have largely focused our work on kinases over-expressed in various cancers and more particularly on the mechanism of activation of AKT- 1 which is involved in numerous regulation pathways and thus in many cancers (Cancer Institute UK). Two papers have been published in PloS Biology, along with a review on the subject. The aim of the project was to unravel at an atomic level the complex activation process of this master kinase. The whole work has been highlighted in England on several internet sites like Yahoo England or Channel Four and in France in the Journal du CNRS. This work has now been extended to the kinase PDK-1, which is one of the major activating factor of the AKT cascade. A paper has been published in Science Signaling.
Â
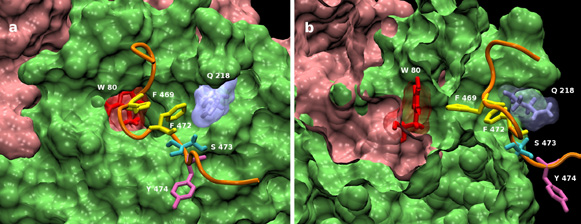
Figure 2: Complete structure of AKT-1: 2 orthogonal views of the Cterm docked on the WT PH/KIN complex : PH in pink, KIN in green, Cterm backbone in orange, W80 in red, F469 & 472 in yellow, S473 in cyan, Y474 in mauve, Q218 in aquablue. The water channel is visible in the center of the left image and on the right it is cut just at its middle by the clipping plane.
We also have considered molecular dynamics of membrane receptors in a full lipidic environment and monitored the drug/receptor interaction. Actually the main interest lies in the GPCR super-family including human dopamin D2 or leukotrien receptors and mainly the opiate receptors in collaboration with Vanderbilt University. A paper has been published in Protein Science. A second has been submitted to Mol. Pharm. This collaboration now extends to the Harvard School of Medecine.
Drug-design & high-troughput in-silico screening
The activity lies at the frontier between biology and chemistry. Starting from a biological problem, we look for small molecules able to interact with protein targets. Virtual screening is performed with pre-filtered chemical databases, or with in-house collections. This approach leads to the discrimination of the best putative ligands which are then synthesized in our group or through collaborations with other teams. A large project for 4 years has been granted by INCA. The subject is Helicase and this is a collaboration with Drs. P. Lestienne and J. Rosenbaum (INSERM U889).
Â
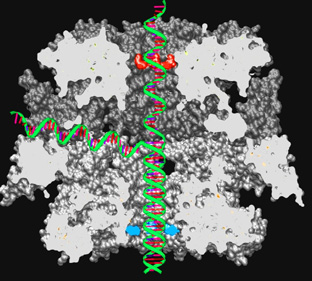
Figure 3: Complete model structure of human Helicase. Reptin is below and pontin above. A double-stranded DNA is entering the protein through a central channel. DNA is unfolded in the center of the complex and then the direct strand gets out by the top pontin pore and the indirect strand is ejected through one of the six lateral channels.
Surface plasmon microscopy
This work is the result of a close collaboration with F. Argoulâs team at ENS Lyon. The goal is to develop a non intrusive tool to detect small variations of the dielectric constant in the vicinity of a metal-dielectric interface. Typical applications range from DNA microarray characterization to cell imaging. Experimentally, the Argoulâs team has shown that dielectric as well as metallic nanoparticles (R > 10 nm) can be detected with this type of microscopy. The technique is based on the interferometric detection of the perturbations induced on the plasmon excitations supported by a thin gold layer (width ~ 50nm). The sample to be imaged is in the vicinity of this layer. Our contribution to this work consists in providing a model that allows a quantitative description of the measurements. The agreement between this theory and the experimental data is good excepted for particle sizes (~ 10 nm) comparable to the scale where the gold deposit cannot be considered as flat. The model also provides with a set of optimal experimental parameters, such as the gold width and the range of incident angles. Overall, the efficiency of plasmon microscopy can be traced back to the amplification by the plasmon excitations of the evanescent waves scattered by the nanoparticles.
Â
Selected publications
- Masters T.A., Calleja V., Armoogum D., Marsh R., Applebee C.J., Laguerre M., Bain A. & Larijani B. (2010). In vivo regulation of 3-phosphoinositide dependent protein kinase 1 (PDK1) activity by homodimerisation. Science Signaling, 3145, ra78.
- Cala O., Pinaud N., Simon C., Fouquet E., Laguerre M., Dufourc E.J. & Pianet I. (2010). The affinity of condensed wine tannins to human saliva proteins is controlled by their conformational preference. A NMR and Molecular Modeling study. FASEB Journal, 24, 4281-4290.
-
Calleja V., Laguerre M., Parker P. J. & Larijani B. (2009). Role of a Novel Phkinase Domain Interface in PKB/Akt Regulations: Structural Mechanism for Allosteric Inhibition. PLoS Biology, 7 (1): 189-200.
-
Oda R., Artzner F., Laguerre M. & Huc I.(2008). Structure of selfassembled chiral nanoribbons and nanotubules revealed in thehydrated state. JACS, 130(44): 14705-14712.
-
Moreau L., Camplo M., Wathier M., Taib N., Laguerre M., Bestel I., Grinstaff M.W. & BarthĂŠlĂŠmy P. (2008). Real Time Imaging of Supramolecular Assembly Formation via Programmed Nucleolipid Recognition. JACS,130(44): 14454-14455.
-
CallĂŠja V., Alcor D., Laguerre M., Park J., Hemmings B., Vojnovic B., Downward J., Parker P.J., Larijani B. (2007). Intra- and inter-molecular interactions of Kinase B define its activation in vivo. PLoS Biology, 5(4): 780-791.
-
Beaurain F., Di Primo C., ToulmĂŠ, J.J., Laguerre M. (2003). Molecular dynamics reveals the stabilizing role of loop closing residues in kissing interactions: comparison between TAR-TAR* and TAR-aptamer. NAR, 31: 4275-4284.
Â
Research team
- Dr. Michel LAGUERRE Team leader
- Dr. Juan ELEZGARAY Research director (DR2, CNRS)
- Dr. Jean DESSOLIN Research officer (CR1, CNRS)
- Dr. Nada TAIB Post-doc (AFM)
- Dr. Marc LAMBLIN Post-doc (ARC)
- Dr. Pramod AKULA BALA Post-doc (ANR)
- Dr. Vincent LEROUX Post-doc (industrial grant, Servier)
- Jean-Michel ARBONA PhD student (MENRT)
- Judith ELKAIM PhD student (MENRT)
- Guillaume NATURALE PhD student (BDI co-financed CNRS/Region)
- Driss BENNANI PhD student (Aquitaine Regional Council)
- Elodie LANDAGARAY Master Student
The team is part of the unit âChimie et Biologie des Membranes et Nanoobjetsâ (CBMN), CNRS/UniversitĂŠ Bordeaux 1/IPB (UMR 5248)
|