| Biomimetic supramolecular chemistry |
|
Dr. Ivan HucDirecteur de recherche (DR1), CNRS This e-mail address is being protected from spambots. You need JavaScript enabled to view itwebsiteTel: +33(0)540002219 BioIvan Huc was born in Besançon, France, in 1969. He studied chemistry at the Ecole Normale Supérieure in Paris, and received his PhD in 1994 from the Université Pierre et Marie Curie (Paris) under the guidance of Dr. C. Rolando (Ecole Normale Supérieure) and Prof. J. Rebek Jr. (Massachusetts Institute of Technology). After a one-year post-doctoral position with Dr. J.-P. Behr at Strasbourg University, he received a CNRS researcher position in the laboratory of Prof. J.-M. Lehn in Strasbourg, where he stayed from 1995 until 1998. Since 1998, he has been a group leader at the Institut Européen de Chimie et Biologie in Bordeaux where he holds a CNRS researchdirector position. In 2008, he started to serve as co-director of the Institute. His current research interests are foldamers and the biomimetic chemistry of peptides and nucleotides. Keywords / Expertise / Techniquesfoldamers, peptidomimetics, helical structures, macrocycles, tertiary structures, peptide-membrane interactions, synthetic organic chemistry, structural studies, X-ray crystallography. SummaryFoldamers – artificial folded molecular architectures – have shifted our knowledge of biopolymer folding in showing that molecular backbones chemically remote from those that nature uses are also able to adopt secondary and tertiary structures. Our group has developed several families of aromatic oligoamides which fold into exceptionally stable, predictable, and tunable conformations. Our current efforts aim at exploring how these aromatic oligoamides may mimic protein tertiary structures and functions, and nucleic acids hybridized architectures, and at investigating their molecular recognition properties and potential biological applications as, for example, ligands for G-quadruplex DNA or protein-protein interaction inhibitors. Activity report
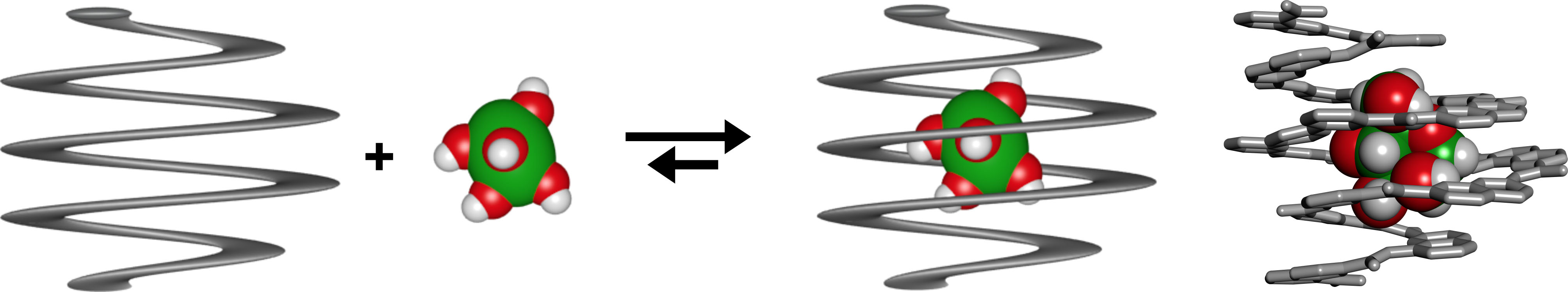
Monosaccharide encapsulation The group routinely prepares a number of heterocyclic amino acid monomers (quinoline, pyridine, naphthyridines, aza-anthracenes…) for the synthesis of aromatic amide foldamers. Monomers notably differ from one another in that they code for helices of various diameters. We have introduced the concept of molecular containers built from the folding of an oligomer into a helix whose diameter is large in the center and small at the ends, and investigated the diastereoselective recognition of chiral guests within these containers. This work recently culminated in the production of specific hosts for monosaccharides (Nat. Chem. 2015). We described the first crystal structures of host-guest complexes involving unsubstituted monosaccharides. Furthermore, these structures allowed to iteratively improve the receptors and reach unprecedented selectivity and atomic scale complementarity for the guests. Foldamer-protein recognition
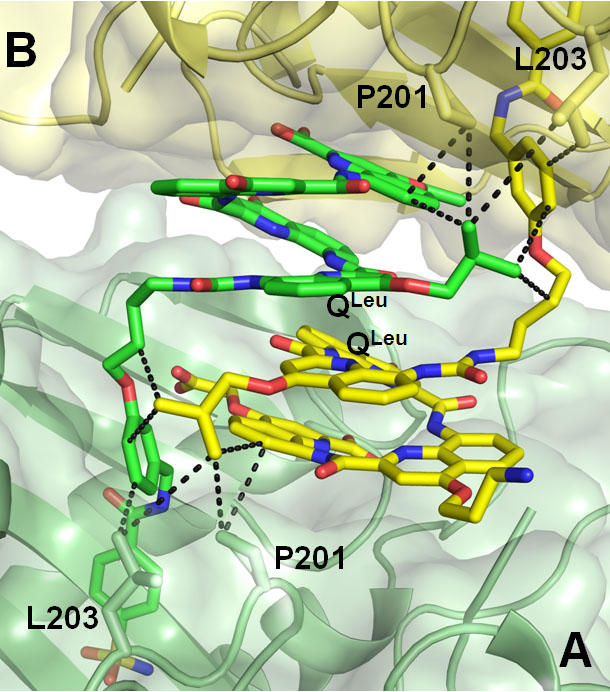
Dr. C. Mackereth at IECB, we identified a foldamer that interacts with a protein surface and characterized the structure of the protein-foldamer complex by single crystal x-ray diffraction (Angew. Chem. Int. Ed. 2014) and in solution (ChemBioChem 2016). We used carbonic anhydrase as a model protein. Foldamers were tethered to the protein surface through the covalent attachment of a nanomolar ligand of the protein active site and foldamer-protein interactions were screened by detecting induced circular dichroism, i.e. the emergence of a preferred foldamer helix handedness induced by interactions with chiral groups at the protein surface. The structure revealed an unexpected dimeric assembly mediated by foldamer-foldamer, foldamer-protein and protein-protein contacts. This ensemble of results represents a major milestone towards the design of medium-sized synthetic ligands for protein surface. Until recently, the synthesis of very long helical aromatic amide foldamers was thought to be limited by steric hindrance associated with stable folded conformations in solution. We showed that this difficulty may be overcome by using pure reagents, relatively high concentrations, and long reaction times (Org. Lett. 2016). Careful identification and elimination of byproducts greatly improve chromatographic purification, giving access to pure products amenable to a segment-doubling synthesis of sequences composed of up to 96 monomers (ca 25 kDa). Compounds as long as 32 units could be prepared on a multigram scale. These results represent a great advance in the availability of foldamers of nanometer size. The physical properties of these compounds are being investigated, including their ability to transport electron and their nanomechanical properties.
Selected publications
Research team
This team is part of the joint research unit “Chimie et Biologie des Membranes et Nanoobjets” (CBMN), CNRS/Univ. Bordeaux/IPB (UMR CNRS 5248) |
2, Rue Robert Escarpit - 33607 PESSAC - France
Tel. : +33 (5) 40 00 30 38 - Fax. : +33 (5) 40 00 30 68



 In a joint effort with biocrystallographers at CBMN (CNRS-Univ. Bordeaux, France), and biomolecular NMR specialist
In a joint effort with biocrystallographers at CBMN (CNRS-Univ. Bordeaux, France), and biomolecular NMR specialist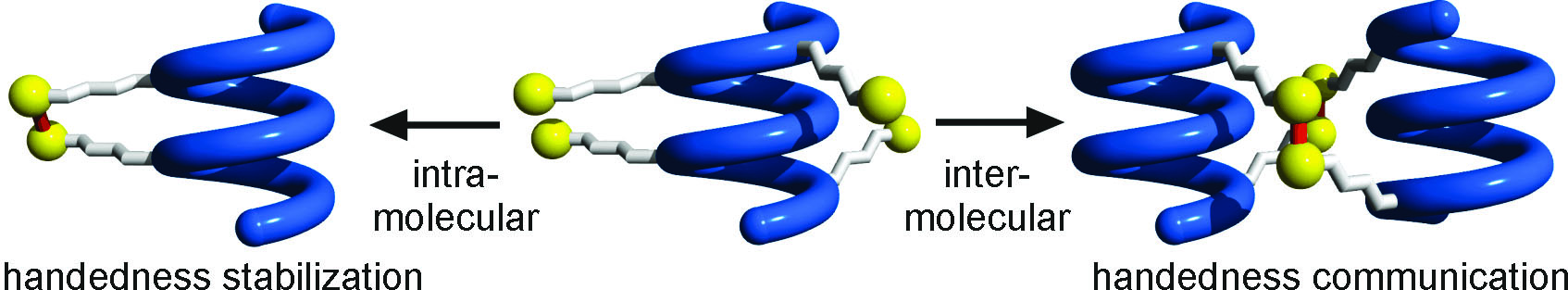 Macrocyclization is a common approach to restrict the conformations available to an otherwise flexible molecular structure. In proteins macrocyclization through disulfide bridges between cysteines far apart in the primary sequence stabilizes folded conformations and contributes to resistance to proteolytic degradation. Disulfide bridge formation was investigated in helical aromatic oligoamide foldamers. Depending on the position of thiol-bearing side chains, exclusive intramolecular or intermolecular disulfide bridging may occur. The two processes are capable of self-sorting, presumably by dynamic exchange. We found that great stabilization of helical structures results, and that helix handedness communication emerge in macrocycles composed of two helices. These results provides an original example of long range conformational coupling that could be useful in synthetic molecular signaling systems and also a powerful tool to design elaborate multi-helical, tertiary-like abiotic architectures (Ang. Chem. Int. Ed., front cover).
Macrocyclization is a common approach to restrict the conformations available to an otherwise flexible molecular structure. In proteins macrocyclization through disulfide bridges between cysteines far apart in the primary sequence stabilizes folded conformations and contributes to resistance to proteolytic degradation. Disulfide bridge formation was investigated in helical aromatic oligoamide foldamers. Depending on the position of thiol-bearing side chains, exclusive intramolecular or intermolecular disulfide bridging may occur. The two processes are capable of self-sorting, presumably by dynamic exchange. We found that great stabilization of helical structures results, and that helix handedness communication emerge in macrocycles composed of two helices. These results provides an original example of long range conformational coupling that could be useful in synthetic molecular signaling systems and also a powerful tool to design elaborate multi-helical, tertiary-like abiotic architectures (Ang. Chem. Int. Ed., front cover).

